Search any question & find its solution
Question:
Answered & Verified by Expert
$\left[\mathrm{Mn}_2(\mathrm{CO})_{10}\right]$ and $\left[\mathrm{Co}_2(\mathrm{CO})_8\right]$ structures have
A. Metal-Metal linkage
B. Terminal CO groups
C. Bridging CO groups
D. Metal in zero oxidation state
Choose the correct answer from the options given below
Options:
A. Metal-Metal linkage
B. Terminal CO groups
C. Bridging CO groups
D. Metal in zero oxidation state
Choose the correct answer from the options given below
Solution:
1234 Upvotes
Verified Answer
The correct answer is:
Only A, B, D
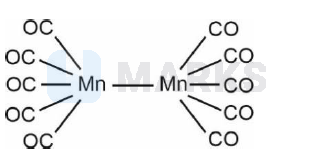
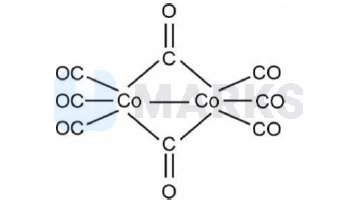
A. Metal-Metal linkage present.
B. Terminal CO groups are present
C. In $\left[\mathrm{Mn}_2(\mathrm{CO})_{10}\right]$ bridging CO groups are not present.
D. Metal is in zero oxidation state.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.