Search any question & find its solution
Question:
Answered & Verified by Expert
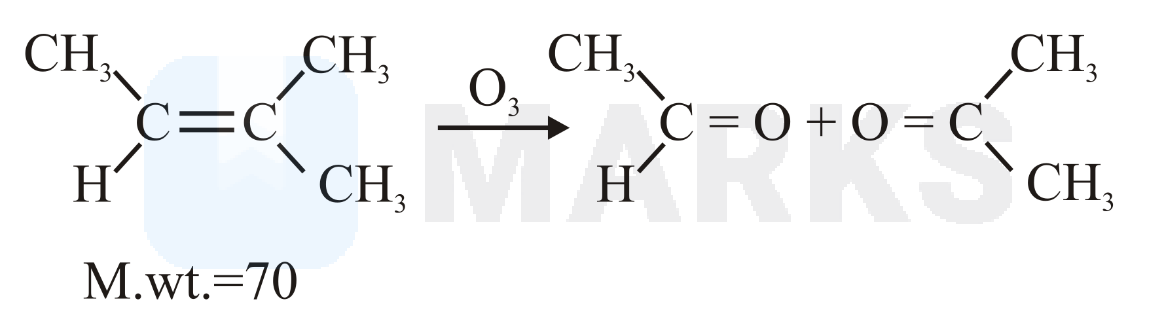
Molar mass of the hydrocarbon (X) which on ozonolysis consumes one mole of per mole of (X) and gives one mole each of ethanal and propanone is ______ g mol–1 (Molar mass of C : 12 g mol–1 , H : 1 g mol–1 )
Solution:
2663 Upvotes
Verified Answer
The correct answer is:
70
When 2-methyl butene-2 is subjected to ozonolysis and then reduced in presence of dimethyl sulphide or Zn dust, it yields acetone and acetaldehyde. Cycloaddition of a molecule of ozone occurs first and then it is cleaved reductively to give aldehyde and ketone.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.