Search any question & find its solution
Question:
Answered & Verified by Expert
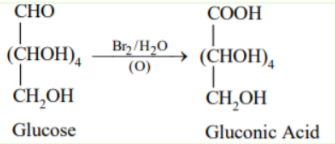
Molecular formula of pentahydroxy acid obtained when glucose is oxidised with $\mathrm{Br}_2$ water is
Options:
Solution:
1000 Upvotes
Verified Answer
The correct answer is:
$\mathrm{C}_6 \mathrm{H}_{12} \mathrm{O}_7$
Glucose on oxidation with bromine water yields gluconic acid $(\mathrm{C} 6 \mathrm{H} 12 \mathrm{O} 7)$. This reaction confirms the presence of an aldehyde group. Because for the formation of gluconic acid, free aldehyde group must be present.

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.