Search any question & find its solution
Question:
Answered & Verified by Expert
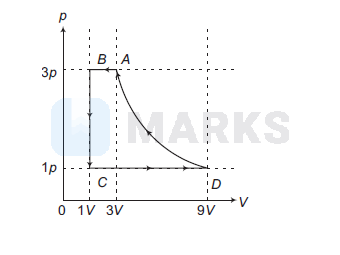
One mole of a monatomic ideal gas is taken through a cycle $A B C D A$ as shown in the $p$-V diagram. Column II gives the characteristics involved in the cycle. Match them with each of the processes given in Column I.

Options:


Solution:
1501 Upvotes
Verified Answer
The correct answer is:
(A) p,q,t, (B) p,r, (C) q,r, (D) s
(A) p,q,t, (B) p,r, (C) q,r, (D) s
Internal energy $\propto T \propto p V$
This is because
$$
U=\frac{n f}{2} R T=\frac{f}{2} p V
$$
Here, $n=$ number of moles
$f=$ degree of freedom
If the product $p V$ increases, then internal energy will increase and if product decreases, the internal energy will decrease.
Further, work is done on the gas, if volume of gas decreases. For heat exchange.
$$
Q=W+\Delta U
$$
Work done is area under $p-V$ graph. If volume increases work done by gas is positive and if volume decreases work done by gas is negative. Further $\Delta U$ is positive if product of $p V$ is increasing and $N U$ is negative, if product of $p V$ is decreasing. If heat is taken by the gas $Q$ is positive and if heat is lost by the gas $Q$ is negative.
Keeping the above points in mind the answer to this question is as under.
(A) $\rightarrow(\mathrm{p}, \mathrm{r}, \mathrm{t})$
(B) $\rightarrow(\mathrm{p}, \mathrm{r})$
(C) $\rightarrow$ (q, s)
(D) $\rightarrow(\mathrm{r}, \mathrm{t})$
Analysis of Question
(i) Calculation wise, question is slightly lengthy. Otherwise question is theory based and simple.
(ii) In process $D A$,
$\begin{array}{rlrl} & & p_A V_A & =p_D V_D \\ \therefore & & T_A & =T_D \\ \text { or } & \Delta U & =0\end{array}$
Further, volume of gas is decreasing. Therefore, work is done on the gas or work done by gas is negative. Therefore, $Q$ is negative or heat is lost.
(iii) This question covers almost all the concepts of first law of thermodynamics.
This is because
$$
U=\frac{n f}{2} R T=\frac{f}{2} p V
$$
Here, $n=$ number of moles
$f=$ degree of freedom
If the product $p V$ increases, then internal energy will increase and if product decreases, the internal energy will decrease.
Further, work is done on the gas, if volume of gas decreases. For heat exchange.
$$
Q=W+\Delta U
$$
Work done is area under $p-V$ graph. If volume increases work done by gas is positive and if volume decreases work done by gas is negative. Further $\Delta U$ is positive if product of $p V$ is increasing and $N U$ is negative, if product of $p V$ is decreasing. If heat is taken by the gas $Q$ is positive and if heat is lost by the gas $Q$ is negative.
Keeping the above points in mind the answer to this question is as under.
(A) $\rightarrow(\mathrm{p}, \mathrm{r}, \mathrm{t})$
(B) $\rightarrow(\mathrm{p}, \mathrm{r})$
(C) $\rightarrow$ (q, s)
(D) $\rightarrow(\mathrm{r}, \mathrm{t})$
Analysis of Question
(i) Calculation wise, question is slightly lengthy. Otherwise question is theory based and simple.
(ii) In process $D A$,
$\begin{array}{rlrl} & & p_A V_A & =p_D V_D \\ \therefore & & T_A & =T_D \\ \text { or } & \Delta U & =0\end{array}$
Further, volume of gas is decreasing. Therefore, work is done on the gas or work done by gas is negative. Therefore, $Q$ is negative or heat is lost.
(iii) This question covers almost all the concepts of first law of thermodynamics.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.