Search any question & find its solution
Question:
Answered & Verified by Expert
One mole of $\mathrm{PCl}_5(\mathrm{~g})$ was heated in a $1 \mathrm{~L}$ closed flask at $500 \mathrm{~K}$. At equilibrium, 0.1 mole of $\mathrm{Cl}_2(\mathrm{~g})$ was formed. What is its $\mathrm{K}_{\mathrm{p}}$ (in atm)?
(Given $\mathrm{R}=0.082 \mathrm{~L} \mathrm{~atm} \mathrm{~mol}^{-1} \mathrm{~K}^{-1}$ )
Options:
(Given $\mathrm{R}=0.082 \mathrm{~L} \mathrm{~atm} \mathrm{~mol}^{-1} \mathrm{~K}^{-1}$ )
Solution:
2147 Upvotes
Verified Answer
The correct answer is:
0.455
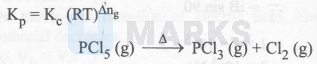
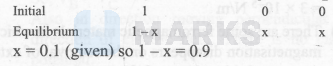
$\begin{aligned} & \Rightarrow \mathrm{K}_{\mathrm{c}}=\frac{\mathrm{x}^2}{1-\mathrm{x}}=\frac{(0.1)^2}{0.9}=0.011 \\ & \text { Thus, } \mathrm{K}_{\mathrm{p}}=(0.011)(0.082 \times 500)^{(2-1)} \\ & =0.451 .\end{aligned}$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.