Search any question & find its solution
Question:
Answered & Verified by Expert
One of the reaction that takes place in producing steel from iron ore is the reduction of iron (II) oxide by carbon monoxide to give iron metal and \(\mathrm{CO}_2\).
\(\mathrm{FeO}(\mathrm{s})+\mathrm{CO}(\mathrm{g}) \rightleftharpoons \mathrm{Fe}(\mathrm{s})+\mathrm{CO}_2(\mathrm{~g}) ;\)
\(K_p=0.265\) atm at \(1050 \mathrm{~K}\)
What are the equilibrium partial pressures of \(\mathrm{CO}\) and \(\mathrm{CO}_2\) at \(1050 \mathrm{~K}\) if the initial partial pressures are : \(\mathrm{p}_{\mathrm{CO}}=1.4\) atm and \(\mathrm{p}_{\mathrm{CO}_2}=0.80 \mathrm{~atm} ?\)
\(\mathrm{FeO}(\mathrm{s})+\mathrm{CO}(\mathrm{g}) \rightleftharpoons \mathrm{Fe}(\mathrm{s})+\mathrm{CO}_2(\mathrm{~g}) ;\)
\(K_p=0.265\) atm at \(1050 \mathrm{~K}\)
What are the equilibrium partial pressures of \(\mathrm{CO}\) and \(\mathrm{CO}_2\) at \(1050 \mathrm{~K}\) if the initial partial pressures are : \(\mathrm{p}_{\mathrm{CO}}=1.4\) atm and \(\mathrm{p}_{\mathrm{CO}_2}=0.80 \mathrm{~atm} ?\)
Solution:
1434 Upvotes
Verified Answer
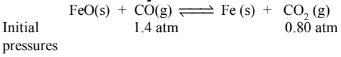
\(\mathrm{Q}_p=\frac{\mathrm{P}_{\mathrm{CO}_2}}{\mathrm{P}_{\mathrm{CO}}}=\frac{0.80}{1.4}=0.571\)
As \(\mathrm{Q}_{\mathrm{p}}>\mathrm{K}_{\mathrm{p}}\), reaction will move in the backward direction, i.e. pressure of \(\mathrm{CO}_2\) will decrease and that of \(\mathrm{CO}\) will increase to attain equilibrium. Hence, if \(\mathrm{P}\) is decrease in pressure of \(\mathrm{CO}_2\), increase in pressure of \(\mathrm{CO}=\mathrm{P}\)
At equilibrium, \(\mathrm{P}_{\mathrm{CO}_2}=(0.80-\mathrm{P})\) atm, \(\mathrm{P}_{\mathrm{CO}}=(1.4+\mathrm{P}) \mathrm{atm}\)
\(\mathrm{K}_{\mathrm{p}}=\frac{\mathrm{P}_{\mathrm{co}_2}}{\mathrm{P}_{\mathrm{co}}} \therefore 0.265=\frac{0.80-\mathrm{P}}{1.4+\mathrm{P}}\)
or \(0.265(1.4+\mathrm{P})=0.80-\mathrm{P}\)
or \(0.371+0.265 \mathrm{P}=0.80-\mathrm{P}\)
or \(1.265 \mathrm{P}=0.429\) or \(\mathrm{P}=0.339\) at equilibrium \(\therefore \quad \mathrm{P}_{\mathrm{CO}}=1.4+0.339=1.739 \mathrm{~atm}\)
and \(\mathrm{P}_{\mathrm{CO}_2}=0.80-0.339=0.461 \mathrm{~atm}\)
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.