Search any question & find its solution
Question:
Answered & Verified by Expert
Order of acidic nature of the following compounds is
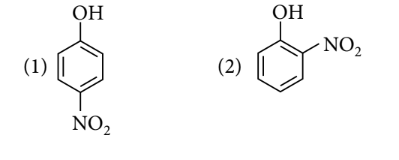
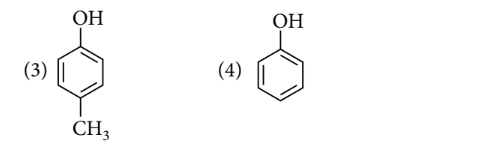
Options:


Solution:
2327 Upvotes
Verified Answer
The correct answer is:
$1>2>4>3$
Electron withdrawing group increases the acidic strength of phenols whereas electron donating group decreases the acidic strength of phenols.
$o$-Nitrophenol is little less acidic than $p$-nitrophenol due to intramolecular $\mathrm{H}$-bonding which makes loss of a proton little difficult.
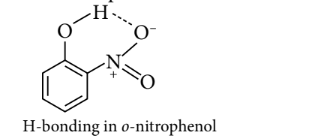
$o$-Nitrophenol is little less acidic than $p$-nitrophenol due to intramolecular $\mathrm{H}$-bonding which makes loss of a proton little difficult.

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.