Search any question & find its solution
Question:
Answered & Verified by Expert
Out of o-nitrophenol and o-cresol which is more acidic?
Solution:
2081 Upvotes
Verified Answer
o-nitrophenol is more acidic than o-cresol. Acidic character of alcohols depends upon the polarity of $\mathrm{O}-\mathrm{H}$ bond. In o-nitrophenol due to $-\mathrm{I}$ and $-\mathrm{R}$ effect of nitro-group electron density decreases in $\mathrm{O}-\mathrm{H}$ bond and polarity increases.
While in o-cresol, due to $+\mathrm{I}$ effect of $-\mathrm{CH}_3$ group electron density of $\mathrm{O}-\mathrm{H}$ bond increases. Therefore, $\mathrm{O}-\mathrm{H}$ bond becomes less polar. Hence, o-nitrophenol is more acidic than o-cresol.
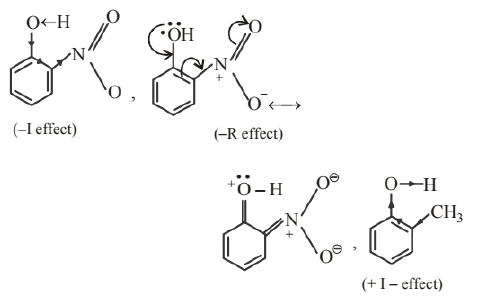
While in o-cresol, due to $+\mathrm{I}$ effect of $-\mathrm{CH}_3$ group electron density of $\mathrm{O}-\mathrm{H}$ bond increases. Therefore, $\mathrm{O}-\mathrm{H}$ bond becomes less polar. Hence, o-nitrophenol is more acidic than o-cresol.

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.