Search any question & find its solution
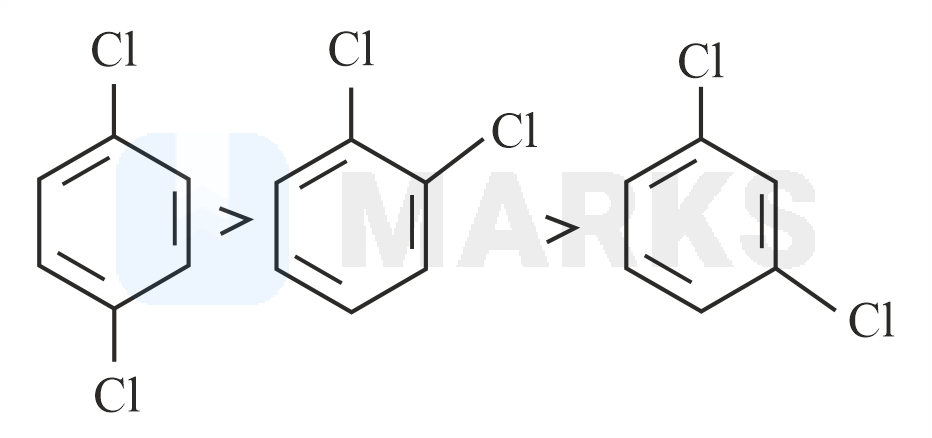
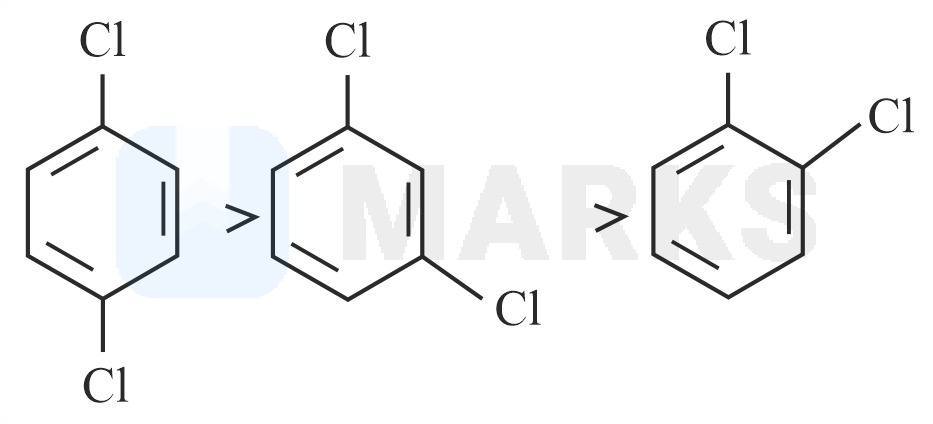
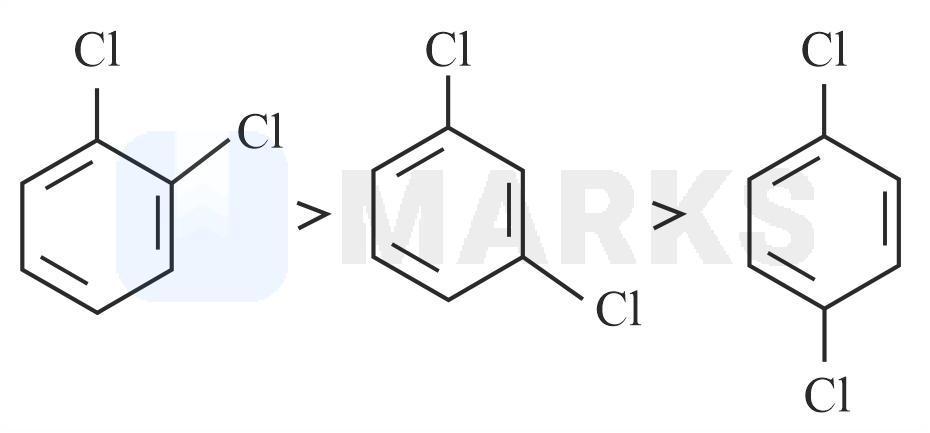
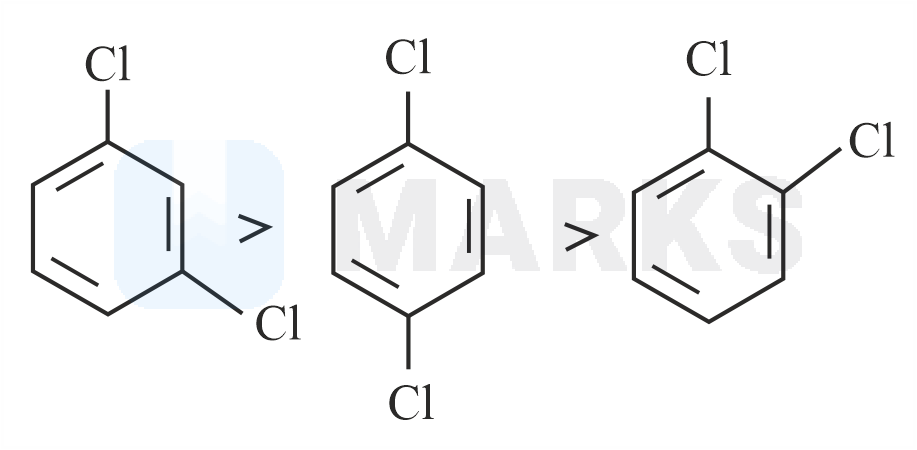
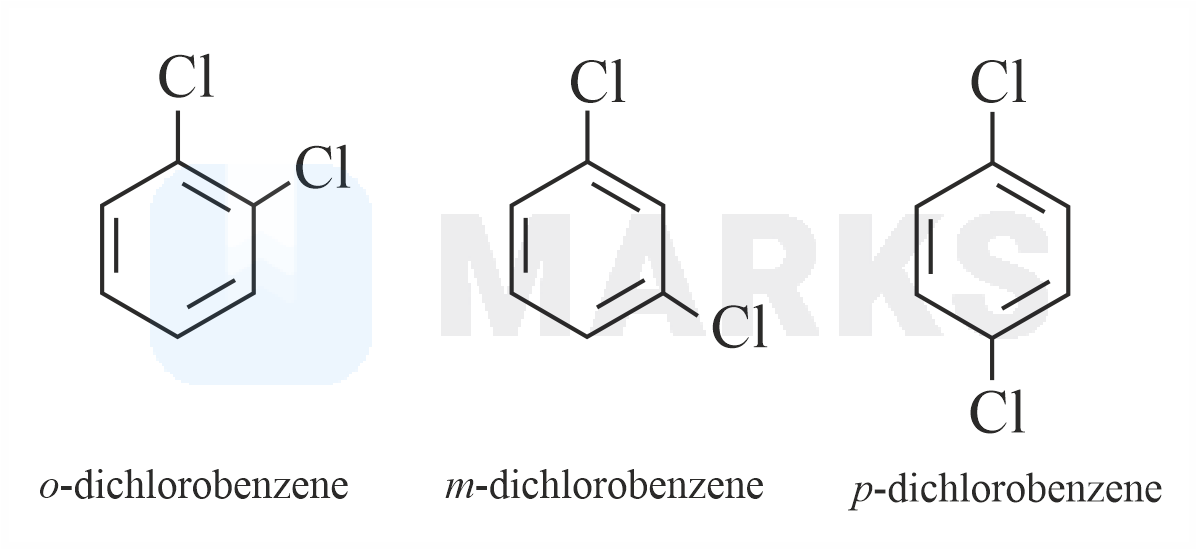
Out of the three isomers of dichlorobenzene, the para-isomer is more symmetrical than the other two isomers, So, it has a more closely packed arrangement of molecules in its crystal lattice. So, para-dichlorobenzene has a higher melting point as compared to ortho and meta Isomers.
Melting point of ortho-dichlorobenzene, meta-dichlorobenzene and para-dichlorobenzene are 256K, 249K, 323K respectively.
Bromo, iodo and polychloro derivatives of hydrocarbons are heavier than water. The density increases with increase in number of carbon atoms, halogen atoms and atomic mass of the halogen atoms.
So, the correct option is D.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.