Search any question & find its solution
Question:
Answered & Verified by Expert
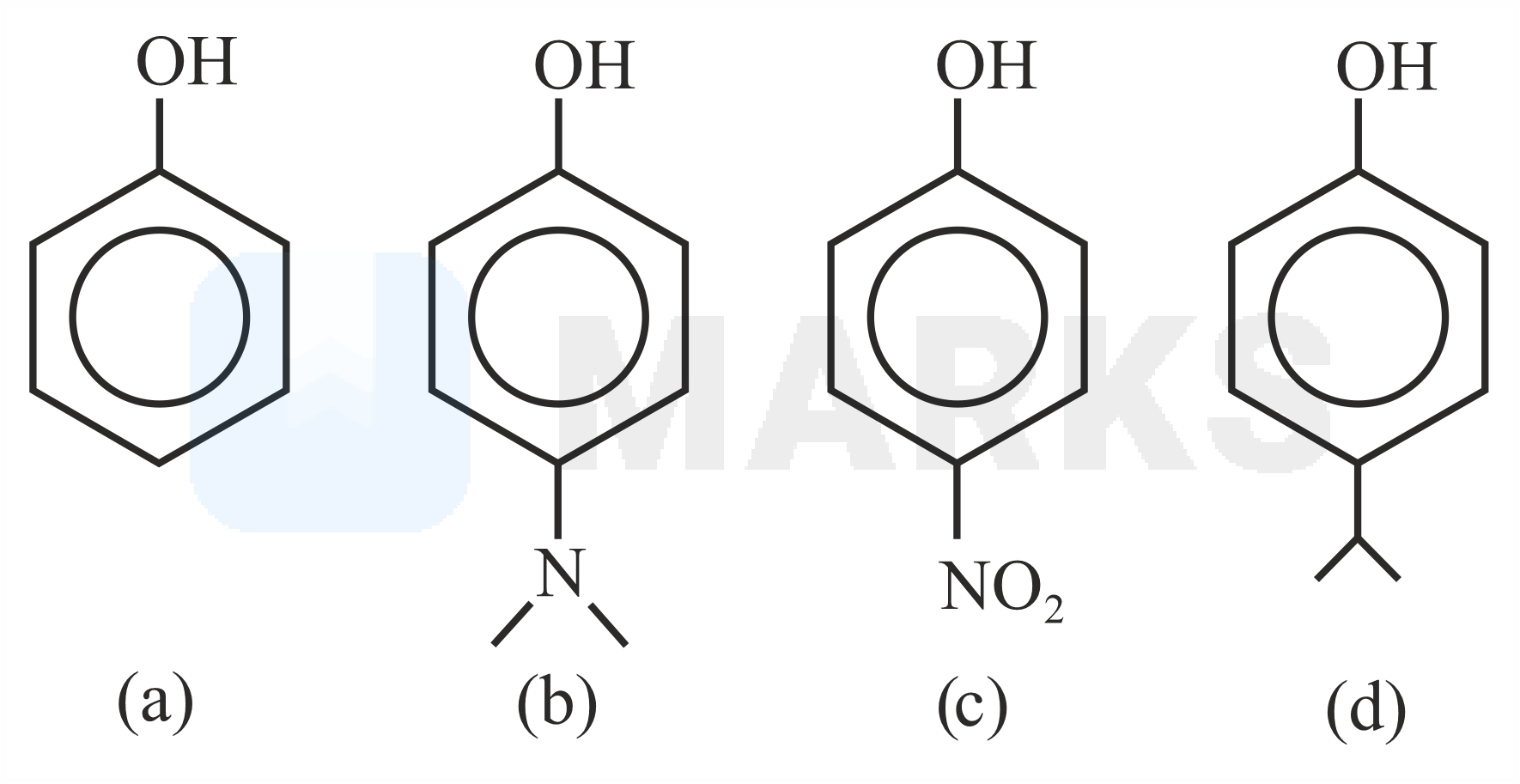
The correct order of values for the following compounds is:
Solution:
1419 Upvotes
Verified Answer
The correct answer is:
b > d > a > c
Electron-withdrawing groups increase the acidic nature of the phenol. Nitro group is electron withdrawing group, hence it is the more acidic compound among the given phenols.
Electron-donating groups decrease the acidic nature where methoxy group is more electron donating, hence less acidic than the iso-propyl group which donates electrons by hyperconjugation effect.
Order of acidic strength is (c) > (a) > (d) > (b)
The order of pKa values are (b) > (d) >(a) > (c).
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.