Search any question & find its solution
Question:
Answered & Verified by Expert
The correct statement regarding the comparison of staggered and eclipsed conformations of ethane, is:
Options:
Solution:
2001 Upvotes
Verified Answer
The correct answer is:
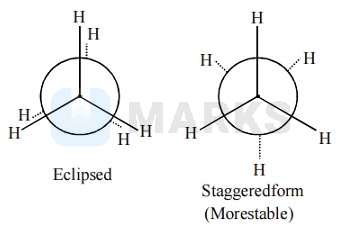
The staggered conformation of ethane is more stable than eclipsed conformation, because staggered conformation has no torsional strain.
The correct statement regarding the comparison of staggered and eclipsed conformations of ethane is the staggered conformation of ethane is more stable than eclipsed conformation, because staggered conformation has no torsional strain.

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.