Search any question & find its solution
Question:
Answered & Verified by Expert
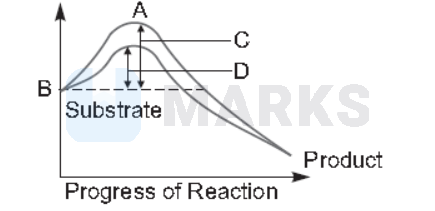
The figure given below shows the conversion of a substrate into product by an enzyme. In which one of the four options (a-d), the components of reaction labelled as $A, B, C$ and $D$ are identified correctly?
 $\begin{array}{|c|c|c|c|c|}
$\begin{array}{|c|c|c|c|c|}
\hline & A & B & C & D \\
\hline \text{(a)} & \text{Potential energy} & \text{Transition state} & \text{Activation energy with enzyme} & \text{Activation energy without enzyme} \\
\hline \text{(b)} & \text{Transition energy} & \text{Potential energy} & \text{Activation energy without enzyme} & \text{Activation energy with enzyme} \\
\hline \text{(c)} & \text{Potential energy} & \text{Transition state} & \text{Activation energy with enzyme} & \text{Activation energy without enzyme} \\
\hline \text{(d)} & \text{Activation energy with enzyme} & \text{Transition state} & \text{Activation energy without enzyme} & \text{Potential energy} \\
\hline
\end{array}$
Options:
 $\begin{array}{|c|c|c|c|c|}
$\begin{array}{|c|c|c|c|c|}
\hline & A & B & C & D \\
\hline \text{(a)} & \text{Potential energy} & \text{Transition state} & \text{Activation energy with enzyme} & \text{Activation energy without enzyme} \\
\hline \text{(b)} & \text{Transition energy} & \text{Potential energy} & \text{Activation energy without enzyme} & \text{Activation energy with enzyme} \\
\hline \text{(c)} & \text{Potential energy} & \text{Transition state} & \text{Activation energy with enzyme} & \text{Activation energy without enzyme} \\
\hline \text{(d)} & \text{Activation energy with enzyme} & \text{Transition state} & \text{Activation energy without enzyme} & \text{Potential energy} \\
\hline
\end{array}$
Solution:
2449 Upvotes
Verified Answer
The correct answer is:
(b)
The amount of energy required to raise the energy of molecules at which chemical reaction can occur is called activation energy. Thus, activation energy is actually the energy required to form the transition state. Enzymes dramatically reduce the activation energy of a reaction, so that most molecules can easily get over the activation energy barrier and quickly turn into product. Simply we can say that activation energy of an enzyme catalysed reaction is lower than that of an uncatalysed reaction.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.