Search any question & find its solution
Question:
Answered & Verified by Expert
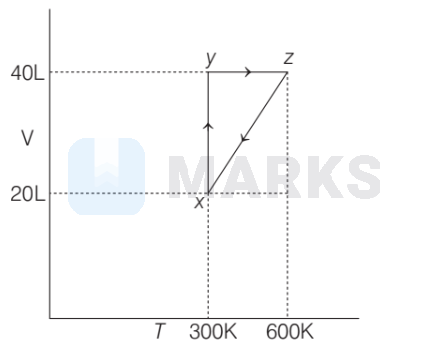
The following graph indicates the system containing 1 mole of gas involving various steps. When it moves from $z$ to $x$, the type of undergoing process is
Options:

Solution:
1534 Upvotes
Verified Answer
The correct answer is:
isobaric
At $x \Rightarrow T_1=300 \mathrm{~K}, V_1=20 \mathrm{~L}$, pressure $=p_1$
At, $z \Rightarrow T_2=600 \mathrm{~K}, V_2=40 \mathrm{~L}$, pressure $=$ ?
According to universal gas law
$\begin{aligned} p V & =n R T \\ p V / T & =n R \quad(n R=\text { constant }) \\ \text { Then, } \quad p_1 V_1 / T_1 & =p_2 V_2 / T_2 \\ \frac{p_1 \times 20}{300} & =\frac{p_2 \times 40}{600} \\ p_1 & =p_2 \\ \Rightarrow \quad \text { i.e. process is isobaric. } & \end{aligned}$
At, $z \Rightarrow T_2=600 \mathrm{~K}, V_2=40 \mathrm{~L}$, pressure $=$ ?
According to universal gas law
$\begin{aligned} p V & =n R T \\ p V / T & =n R \quad(n R=\text { constant }) \\ \text { Then, } \quad p_1 V_1 / T_1 & =p_2 V_2 / T_2 \\ \frac{p_1 \times 20}{300} & =\frac{p_2 \times 40}{600} \\ p_1 & =p_2 \\ \Rightarrow \quad \text { i.e. process is isobaric. } & \end{aligned}$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.