Search any question & find its solution
Question:
Answered & Verified by Expert
The geometries of $\left[\mathrm{Ni}(\mathrm{CO})_4\right],\left[\mathrm{PtCl}_4\right]^{2-}$ and $\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_6\right]^{3+}$ respectively are
Options:
Solution:
1102 Upvotes
Verified Answer
The correct answer is:
tetrahedral, square planar and octahedral
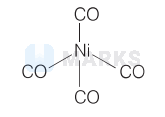
$\left[\mathrm{Ni}(\mathrm{CO})_4\right]$
Ground state electronic configuration $=3 d^8 4 s^2$
$\mathrm{CO}$ is strong field ligand, so it pushes $10 e^{-}$to $3 d$-orbital. The hybridisation of $\left[\mathrm{Ni}(\mathrm{CO})_4\right]$ is $s p^3$ and geometry is tetrahedral.
$\left[\mathrm{PtCl}_4\right]^{2-} \rightarrow$ Square planar. A strong repulsion with the electron and ligand take place in between platinum which leads to strong field splitting due to which square planar geometry is more stabilised than tetrahedral shape.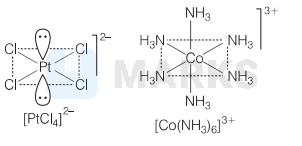
$\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_6\right]^{3+}$ is octahedral that involves $d^2 s p^3$ hybridisation.
Ground state electronic configuration $=3 d^8 4 s^2$
$\mathrm{CO}$ is strong field ligand, so it pushes $10 e^{-}$to $3 d$-orbital. The hybridisation of $\left[\mathrm{Ni}(\mathrm{CO})_4\right]$ is $s p^3$ and geometry is tetrahedral.

$\left[\mathrm{PtCl}_4\right]^{2-} \rightarrow$ Square planar. A strong repulsion with the electron and ligand take place in between platinum which leads to strong field splitting due to which square planar geometry is more stabilised than tetrahedral shape.

$\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_6\right]^{3+}$ is octahedral that involves $d^2 s p^3$ hybridisation.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.