Search any question & find its solution
Question:
Answered & Verified by Expert
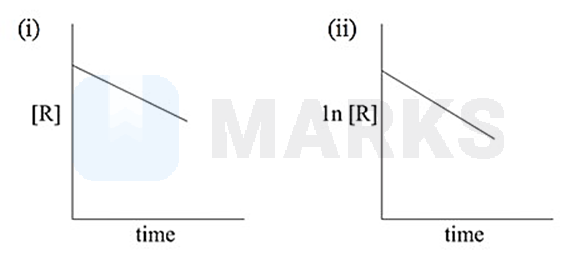
The given plots represent the variation of the concentration of a reaction R with time for two different reaction (i) and (ii). The respective orders of the reactions are
Options:

Solution:
1355 Upvotes
Verified Answer
The correct answer is:
0, 1
Intergrated rate law for zero order reactions is or or ( given)
On comparing with
slope for graph between (R) and t with intercept
first graph is of zero order reactions.
Integrated rate law for first order reaction is
On comparing with
Slope for graph between 1n (R) and t with intercept 1n
Second graph is of first order
On comparing with
slope for graph between (R) and t with intercept
first graph is of zero order reactions.
Integrated rate law for first order reaction is
On comparing with
Slope for graph between 1n (R) and t with intercept 1n
Second graph is of first order
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.