Search any question & find its solution
Question:
Answered & Verified by Expert
The hybridizations of $\mathrm{Ni}(\mathrm{CO})_{4}$ and $\mathrm{Cr}\left(\mathrm{H}_{2} \mathrm{O}\right)_{6}^{2+}$, respectively, are
Options:
Solution:
2250 Upvotes
Verified Answer
The correct answer is:
$\mathrm{sp}^{3}$ and $\mathrm{d}^{2} \mathrm{sp}^{3}$
$\mathrm{Ni}(\mathrm{CO})_{4}$
$\mathrm{Ni}^{0}=3 \mathrm{~d}^{8} 4 \mathrm{~s}^{2}$
By effect of S.F.L. $\mathrm{CO}$.
$\left[\mathrm{Cr}\left(\mathrm{H}_{2} \mathrm{O}\right)_{6}\right]^{+2} \quad \mathrm{Cr}^{+2}=3 \mathrm{~d}^{4} 4 \mathrm{~s}^{0} 4 \mathrm{p}^{0}$
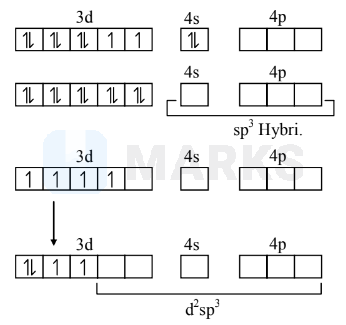
$\mathrm{Ni}^{0}=3 \mathrm{~d}^{8} 4 \mathrm{~s}^{2}$
By effect of S.F.L. $\mathrm{CO}$.
$\left[\mathrm{Cr}\left(\mathrm{H}_{2} \mathrm{O}\right)_{6}\right]^{+2} \quad \mathrm{Cr}^{+2}=3 \mathrm{~d}^{4} 4 \mathrm{~s}^{0} 4 \mathrm{p}^{0}$

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.