Search any question & find its solution
Question:
Answered & Verified by Expert
The increasing order of basicity of the following compounds is :
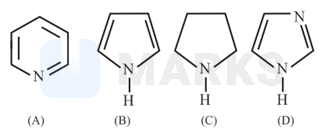
Solution:
2554 Upvotes
Verified Answer
The correct answer is:
The basic nature of amines can be explained on the basis hybridisation. The basic nature order is . If the lone pair on nitrogen involved in resonance, then it is less basic than others. In the molecules A, B and D, the nitrogen is hybridised. In the pyrrole molecule the lone pair on nitrogen involving in conjugation to get aromaticity. Hence, C is most basic and B is least basic.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.