Search any question & find its solution
Question:
Answered & Verified by Expert
The increasing order of the reactivity of the following with is:
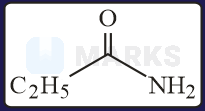
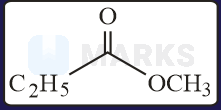
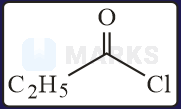
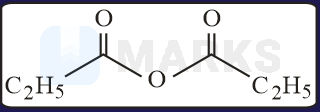
Solution:
2380 Upvotes
Verified Answer
The correct answer is:
In the mentioned structures will get added to the carbon having slight positive charge.
Presence of increases the positive charge whereas an electron donating group like decreases the tendency.
Rate of reaction or group
Rate of reaction and
Hence, the correct order will be: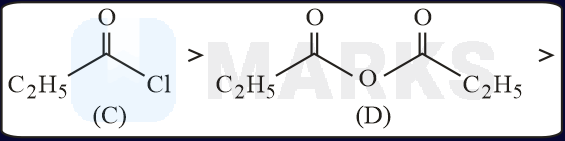
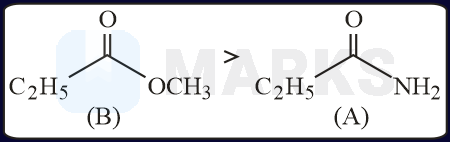
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.