Search any question & find its solution
Question:
Answered & Verified by Expert
The isotherms of a gas are shown below :
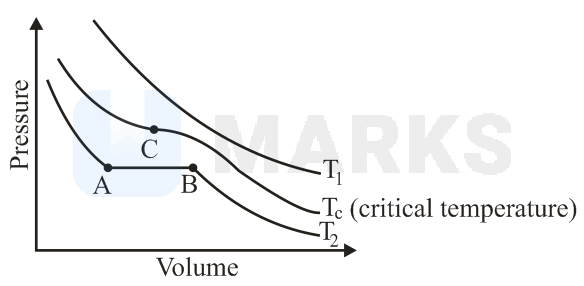
Among the following,
(i) At , the gas cannot be liquefied
(ii) At point , liquid starts to appear at
(ii) is the highest temperature at which the gas can be liquefied
(iv) At point , a small increase in pressure condenses the whole system to a liquid
The correct statements are :
Solution:
2765 Upvotes
Verified Answer
The correct answer is:
(i), (ii), (iii) and (iv)
Since , the gas cannot be liquefied at , is the highest temperature at which the gas can be liquefied.
At temperature , liquid starts to appear at point , however a small increase in pressure at point condenses the whole system to liquid.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.