Search any question & find its solution
Question:
Answered & Verified by Expert
The $K_{\mathrm{H}}$ value ( $\mathrm{K}$ bar) of argon (I), carbondioxide (II), formaldehyde (III) and methane (IV) are respectively 40.3, 167, $1.83 \times 0^{-5}$ and $0.413$ at $298 \mathrm{~K}$. The increasing order of solubility of gas in liquid is
Options:
Solution:
1209 Upvotes
Verified Answer
The correct answer is:
I $ < $ II $ < $ IV $ < $ III
Higher is the $K_{\mathrm{H}}$ value, less is the solubility of the gas.
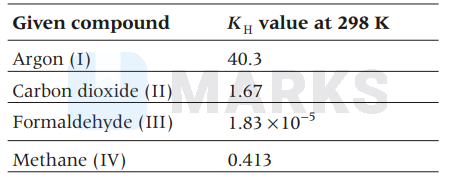
Thus, the increasing order of solubility of gas in liquid is I < II < IV < III.

Thus, the increasing order of solubility of gas in liquid is I < II < IV < III.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.