Search any question & find its solution
Question:
Answered & Verified by Expert
The oxidation state $(n)$ and coordination number $(\mathrm{CN})$ of $\mathrm{Al}$ and number of valence electrons around $\mathrm{Al}(\mathrm{N})$ in $\mathrm{Al}_2 \mathrm{Cl}_6$ are respectively
Options:
Solution:
1475 Upvotes
Verified Answer
The correct answer is:
$3,4,8$
$$
\text { Structure of } \mathrm{Al}_2 \mathrm{Cl}_6 \text { is }
$$
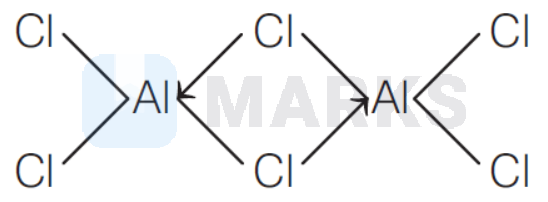
Therefore,
(i) Oxidation state $(x)$ of 'Al' in $\mathrm{Al}_2 \mathrm{Cl}$ :
$$
=2 x-6=0 \text { or } x=\{+\rangle
$$
(ii) Coordination number of $\mathrm{Al}$ in $\mathrm{Al}_2 \mathrm{Cl}_5$ is four (4), it is bonded to 4 chlorine atoms in $\mathrm{Al}_2 \mathrm{Cl}_5$.
(iii) Number of valence electrons around Al in
$$
\mathrm{Al}_2 \mathrm{Cl}_6=2 \times 4=8
$$
Hence, option (2) is the correct answer.
\text { Structure of } \mathrm{Al}_2 \mathrm{Cl}_6 \text { is }
$$

Therefore,
(i) Oxidation state $(x)$ of 'Al' in $\mathrm{Al}_2 \mathrm{Cl}$ :
$$
=2 x-6=0 \text { or } x=\{+\rangle
$$
(ii) Coordination number of $\mathrm{Al}$ in $\mathrm{Al}_2 \mathrm{Cl}_5$ is four (4), it is bonded to 4 chlorine atoms in $\mathrm{Al}_2 \mathrm{Cl}_5$.
(iii) Number of valence electrons around Al in
$$
\mathrm{Al}_2 \mathrm{Cl}_6=2 \times 4=8
$$
Hence, option (2) is the correct answer.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.