Search any question & find its solution
Question:
Answered & Verified by Expert
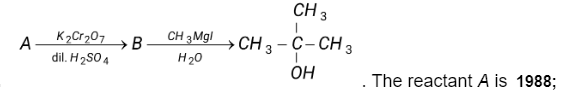 The reaction is:
The reaction is: Solution:
1914 Upvotes
Verified Answer
The correct answer is:
$\mathrm{CH}_3 \mathrm{COCH}_3$
The reaction shows a two-step process:
- In the first step, potassium dichromate ($\mathrm{K} 2 \mathrm{Cr} 2 \mathrm{O}_7)$) oxidizes the alcohol (reactant A) to a ketone or aldehyde in the presence of dilute sulfuric acid (H2SO4).
- In the second step, the ketone or aldehyde is reduced to a primary alcohol by methylmagnesium iodide (CH3Mgl) in water ($\mathrm{H} 2 \mathrm{O})$.
The reactant $\mathrm{A}, \mathrm{C} 2 \mathrm{H} 5 \mathrm{OH}$, is ethanol, a primary alcohol. The product of the first step is acetaldehyde, a ketone. The second step reduces the ketone to propanol, another primary alcohol.
- In the first step, potassium dichromate ($\mathrm{K} 2 \mathrm{Cr} 2 \mathrm{O}_7)$) oxidizes the alcohol (reactant A) to a ketone or aldehyde in the presence of dilute sulfuric acid (H2SO4).
- In the second step, the ketone or aldehyde is reduced to a primary alcohol by methylmagnesium iodide (CH3Mgl) in water ($\mathrm{H} 2 \mathrm{O})$.
The reactant $\mathrm{A}, \mathrm{C} 2 \mathrm{H} 5 \mathrm{OH}$, is ethanol, a primary alcohol. The product of the first step is acetaldehyde, a ketone. The second step reduces the ketone to propanol, another primary alcohol.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.