Search any question & find its solution
Question:
Answered & Verified by Expert
What is the formal charge on carbon atom in $\mathrm{CO}_3^{2-}$ ion?
Options:
Solution:
1178 Upvotes
Verified Answer
The correct answer is:
zero
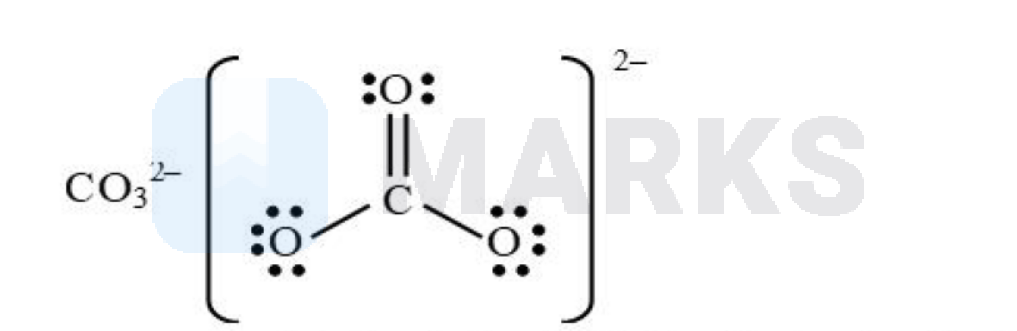
$$
\begin{aligned}
& \text { Formal charg } e=\left[\begin{array}{c}
\text { Total no.of valence } \\
\mathrm{e}^{-} \text {in freestate }
\end{array}\right]-\left[\begin{array}{c}
\text { Total no.oflone } \\
\text { pair electrons }
\end{array}\right]-\frac{1}{2}\left[\begin{array}{c}
\text { no.of bonding electrons } \\
\text { on that particular atom }
\end{array}\right] \\
& =4-0-\frac{1}{2} \times 8=0 \\
&
\end{aligned}
$$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.