Search any question & find its solution
Question:
Answered & Verified by Expert
Which of the following is more basic than aniline?
Options:
Solution:
2654 Upvotes
Verified Answer
The correct answer is:
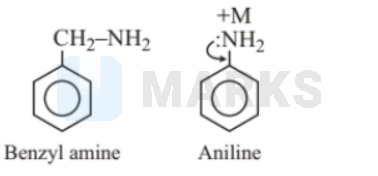
benzylamine
Benzyl amine is more basic than aniline because the benzyl group is an electron-donating group on account of the + I effect. So, it is capable of increasing the electron density of $\mathrm{N}$ of the $-\mathrm{NH}_2$ group. Thus, because of the higher electron density, the rate of donation of a free pair of electrons is increased, i.e., the basic character is higher. Phenyl and nitro groups are electron attractive groups, so they are able to decrease the electron density of $\mathrm{N}$ of $-\mathrm{NH}_2$ group. Hence, they are less basic than aniline.
A Caution
Benzylamine has localised lone pair therefore it is more basic.

A Caution
Benzylamine has localised lone pair therefore it is more basic.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.