Search any question & find its solution
Question:
Answered & Verified by Expert
Why are aryl halides less reactive towards nucleophilic substitution reactions than alkyl halides? How can we enhance the reactivity of aryl halides?
Solution:
1957 Upvotes
Verified Answer
Aryl halides are less reactive than alkyl halides towards nucleophilic substitution reaction due to the following reasons
(i) The C-Cl bond in haloarenes acquires partial double bond character due to resonance stabilisation which strengthen the $\mathrm{C}-\mathrm{Cl}$ bond and makes them less reactive towards nucleophilic substitution reaction.
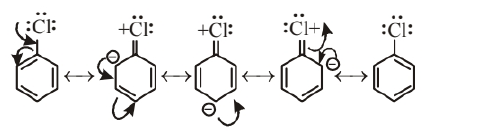
(ii) In haloarenes carbon attached to halogen is $s p^2$ hydridised and is more electronegative. This $s p^2-$ hybridised carbon make $\mathrm{C}-\mathrm{Cl}$ bond shorter than $\mathrm{C}-\mathrm{Cl}$ bond of haloalkanes. Shorter bonds are difficult to break than longer one. Therefore, haloarenes are less reactive than haloalkanes.
(iii) Because of the repulsion between the nucleophile and electron rich arenes, aryl halides are less reactive than alkyl halides.
The reactivity of aryl halides can be increased by the presence of an electron withdrawing group at ortho and para positions. Electron withdrawing group enhance reactivity at ortho and para positions and has no effect on reactivity at meta position.
(i) The C-Cl bond in haloarenes acquires partial double bond character due to resonance stabilisation which strengthen the $\mathrm{C}-\mathrm{Cl}$ bond and makes them less reactive towards nucleophilic substitution reaction.

(ii) In haloarenes carbon attached to halogen is $s p^2$ hydridised and is more electronegative. This $s p^2-$ hybridised carbon make $\mathrm{C}-\mathrm{Cl}$ bond shorter than $\mathrm{C}-\mathrm{Cl}$ bond of haloalkanes. Shorter bonds are difficult to break than longer one. Therefore, haloarenes are less reactive than haloalkanes.
(iii) Because of the repulsion between the nucleophile and electron rich arenes, aryl halides are less reactive than alkyl halides.
The reactivity of aryl halides can be increased by the presence of an electron withdrawing group at ortho and para positions. Electron withdrawing group enhance reactivity at ortho and para positions and has no effect on reactivity at meta position.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.